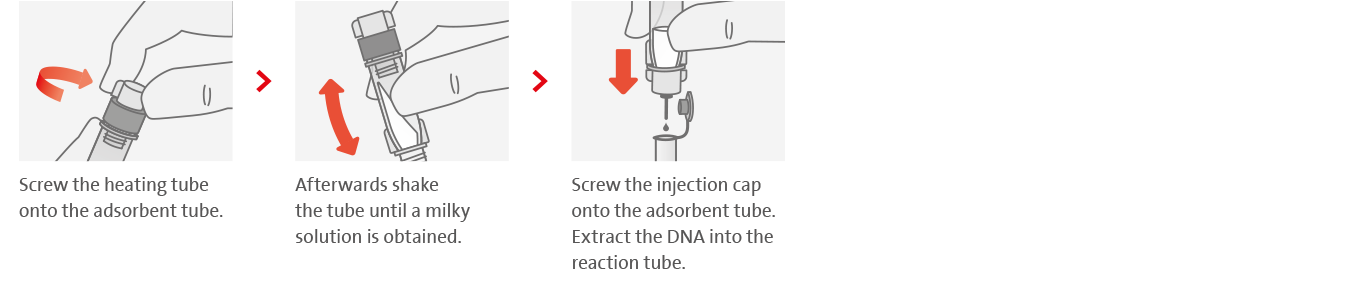
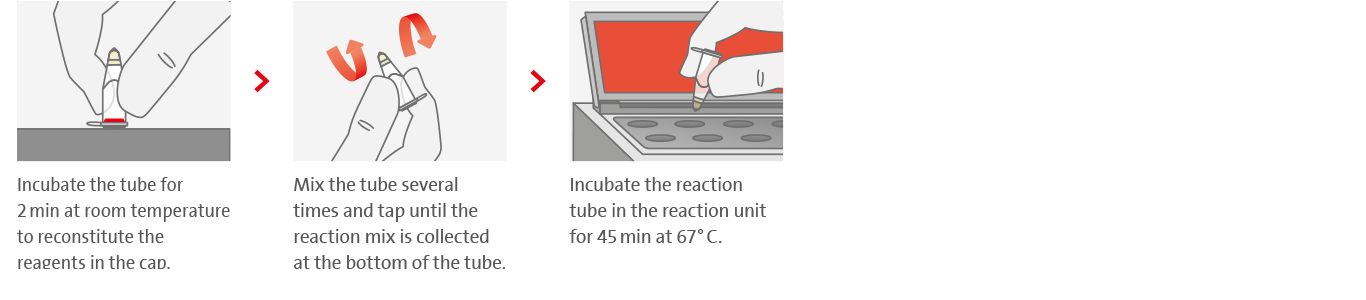
Tuberculosis remains one of the major causes of death worldwide. In 2020, there were an estimated 9.9 million TB cases according to the WHO, of which about 1.5 million led to death. The majority of these cases occur in low- and middle-income countries. Most frightening, however, is the fact that of the estimated 9.9 million cases, about 4.1 million remain undetected. Gaps between the estimated number of new cases and the number actually reported are due to underreporting of cases detected and underdiagnosis or misdiagnosis. The reasons for that are manifold.
Institute Pasteur International
Dr. Valerie Flore Donkeng Donfack